|
Other terms: atomic mass of Nitrogen dioxide, molar mass of Nitrogen dioxide, molecular mass, Use the mole ratio and empirical formula to understand the limits of the reactants. Percent yield calculator which can help you apply this to actual experiments. This project started with as a molar mass calculator for chemical reactions. Note that the calculator assumes a pure substance - if you'reĪware of dilution or impurities, make appropriate adjustments for the molarity of a given substance. The calculator takes the elemental composition of the compound and weighs the elements to get an 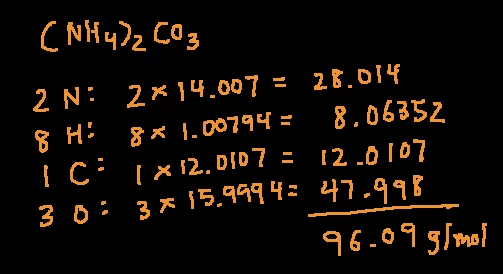
The molar mass of a chemical compound based on the compound's empirical formula. Need to know the atomic mass of a Nitrogen dioxide molecule? Our molar mass calculator uses the periodic table and the chemical formula to solve for of atoms x molar mass of an atomĪnswer the molar mass of an ozone molecule is 48 g/mol.Solving for the atomic mass of Nitrogen dioxide (NO2) whatever the data is given to you, the answer would always be equal to the atomic mass (given in literature) of the substance. Hence, the molar mass is always equal to the atomic mass of a substance. Number of moles present in 30 grams of sodium = mass/molar mass (present in literature) Example # 1Ĭalculate the molar mass of 30 g sodium metal. following is the example to prove that molar mass is always equal to the atomic mass of a substance. It would tell you the molar mass of the given substance. Simply enter the molecular formula or the name of the substance and press the “calculate” button. It is easier to use a molar mass calculator to avoid this hassle. The molar mass of a molecule with the same atoms = no. Molar mass = mass / number of moles of a substance But if you are given a certain number of grams of a substance and asked to find its molar mass then there is a formula to calculate it (it would always be equal to the atomic mass of that substance). In the case of elements, it is given in the literature while in the case of elements and compounds, you can calculate it using the simple fragments method. Molar mass is the mass per mole of a substance and is written as “M”. For example, the atomic mass of fluorine is 19 so the molar mass of fluorine is 19 g/mol. Molar mass is equal to the atomic mass of an element and is expressed in grams per mole (g/mol). In the case of a new compound, it is first converted into fragments and then each fragment is studied separately. Molar mass plays an important role in the structure elucidation step. 
The structure of the element / compound is designed using all this data. Then comes the structure elucidation of the element where its empirical and molecular formula is predicted. When a new element or a compound is discovered then first of all its physical and chemical properties are studied. A molar mass calculator is used to calculate the molar mass of a given element. Hence, we can say that the molar mass of each element is also specific and is stated in the literature. Molar mass is the weight of one mole of an element and as we know that one mole of every element contains a specific number of atoms of that element. It is different for all elements and is used in the identification of an element or a compound. Every substance that is stated in the periodic table exhibits a specific molar mass.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
